MCB TRANSCRIPT

Meiosis as Development: How Translation Drives Differentiation
Meiosis may have evolved from mitosis, but gamete formation is far more than a specialized type of cell division. “Meiosis is a kind of differentiation,” says MCB Assistant Professor Gloria Brar, who was an MCB undergraduate and is happy to be back. “It makes different cell types that can’t go back to what they were before. It’s a metamorphosis. The cell structure changes dramatically.”

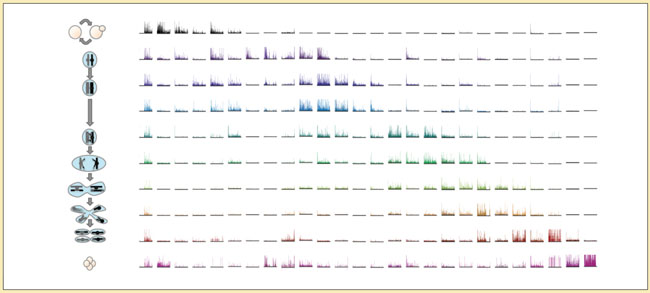
Brar wants to know how changes in gene expression drive structural and functional changes in cells during meiosis, and is starting to find out thanks to the ribosome profiling technique developed by MCB colleague Nicholas Ingolia. “Ribosome profiling gives an instantaneous look at which proteins the cell is making,” she says.
Ribosome profiling let Brar track protein synthesis during yeast meiosis, revealing complex gene expression patterns. Strikingly, the Unfolded Protein Response (UPR) — which is essential to embryonic development in mammals — switches on and off during meiosis. And this UPR modulation appears to be in sync with structural changes in the endoplasmic reticulum, where many proteins are made.
Today, Brar is studying whether changes in the endoplasmic reticulum are important for development, and whether UPR activation during yeast meiosis is similar to its regulation in other developmental processes such as immune cell differentiation.
Another discovery from ribosome profiling is that during meiosis, short sequences of so-called untranslated regions of mRNAs are in fact frequently translated. “This was a big surprise,” Brar says. “It fundamentally challenges the simple model of translation.” Now she’s studying what makes translation different during meiosis. Possibilities include structural changes that let ribosome proteins recognize mRNA sequences that usually aren’t translated.
Ribosome profiling also revealed the translation of so-called non-coding RNAs. “This was an even bigger surprise — it’s transformative to the way we think about genome coding,” she says. It turns out that during yeast meiosis, what initially appeared to be non-coding RNAs are actually translated into 2,000 little proteins. At fewer than 100 amino acids, such proteins had previously been defined as too small to be functional.
“As biologists, we sometimes get caught up in models that we’ve been used to for a long time,” she observes. There are only 6,000 yeast genes for big proteins, making the little proteins a huge new pool, and Brar is knocking them down one by one to discover what they do during meiosis.
Brar’s out-of-the-box findings have sparked interest widely. Last spring, she was selected as one of Cell’s 40 under 40 outstanding young life science researchers from around the world.
Fun Fact: Gloria Brar was MCB's 2002 Departmental Citation winner — the top award for a graduating undergraduate!




