MCB TRANSCRIPT
Gametogenesis and Aging: How to Make the Old New Again
MCB Assistant Professor Elçin Ünal studies the earliest part of life — gamete formation — to learn how to counteract aging later on. This may seem paradoxical but the two are linked. Cells accumulate defects as they age, yet gametes made from old cells somehow shed this damage. “The progeny of a 20 year old man and a 70 year old are similar,” she says. “Why?”

As a post-doc, Ünal found that the same holds for spores from yeast cells so old they were about to die. Even though the old cells were full of genetic and morphological defects, their spores were not. Moreover, cells from these “old” spores lived as long as those from “young” spores. “Gametogenesis resets the lifespan,” she says.
In addition, Ünal discovered that a transcription factor specific to gametogenesis (Ndt80) also makes old yeast cells younger, removing damage and extending their lifespan. “Can we use the rejuvenation processes of gametogenesis to reverse aging in mammals?” she asks. As a first step toward finding out, she’s studying a similar transcription factor (CEP-1) in the worm Caenorhabditis elegans.
Intriguingly, the yeast and worm transcription factors are similar to proteins in the p53 family, which suppress tumors in vertebrates, and the worm transcription factor is also similar to one involved in oocyte quality control in mice. Taken together, this suggests that these tumor suppressors evolved from proteins that originally had a different function.
“We favor the notion that the ancient function of the p53 family was quality control during gametogenesis, and that this was later modified to suppress tumors in response to DNA damage and other cellular stresses,” Ünal says.
Another side of gametogenesis and aging is that as women get older, meiosis — the type of cell division that creates sex cells — is more likely to go wrong. “After age 35, there’s an incredible increase in embryos with imbalanced chromosomes,” Ünal says, adding that monosomy and trisomy are usually lethal and that the latter can lead to Down syndrome.
To learn how this happens, she’s using live imaging of single yeast cells during meiosis. “We’re observing chromosome segregation and the conditions when it goes awry,” she says. For example, the process of protecting centromeric cohesion — which links sister chromatids — is susceptible to maternal age.
Ünal is pleased to be part of MCB. “I’m very fortunate,” she says. “It’s an exceptional place with outstanding scientists, who are also amazing colleagues. It’s truly inspiring to be here.”
This year, Ünal was selected as one of 22 Pew Scholars in the Biomedical Sciences, which supports young health sciences researchers with innovative approaches and ideas.
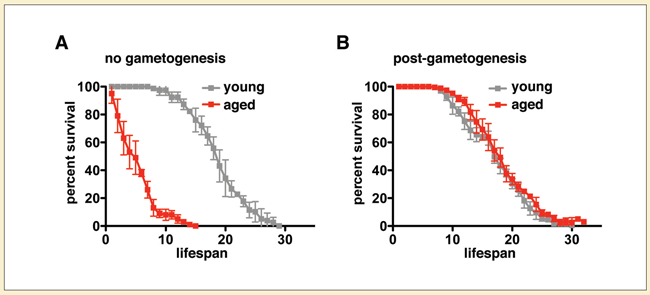
(B) Gametes from old and young cells have the same lifespan.




