Yvette Fisher

Assistant Professor (Affiliated) of Cell Biology, Development and Physiology
Lab Homepage: https://www.fisherlab.science/Research Interests
Current Projects
Synaptic plasticity of sensory inputs
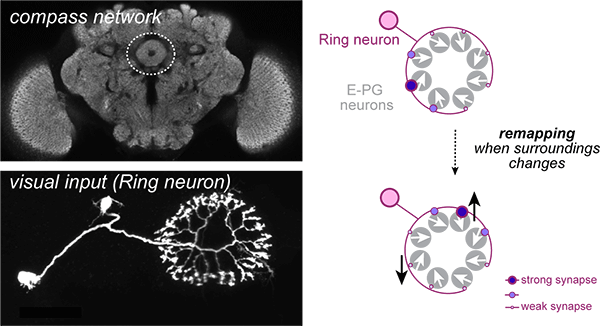
How does experience change synaptic connections in order to flexibly store information? Despite decades of important work on synaptic plasticity, it remains a challenge to directly relate changes in synaptic connections to the emergence of spatial memories. Our previous work suggests that within the compass network, Ring→E-PG synapses store the mapping between sensory cues and heading direction. We use in vivo electrophysiology, genetic manipulations, and virtual reality during 2-photon imaging to directly relate synaptic changes to the formation of new spatial memories. This multi-faceted approach will allow us to understand how the precise logic of synaptic plasticity shapes learning during exploration.
Context-dependent neuromodulation
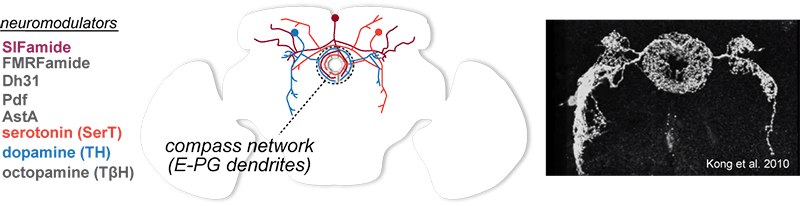
How does context shape our sense of direction? Navigational circuits experience different constraints depending on context. For example, we have different expectations about our upcoming movements through the world when riding a bicycle versus walking slowly. In this project we explore the hypothesis that distinct neuromodulatory systems serve toswitch the compass network into different operating modes depending on context.
Homeostasis of compass network properties
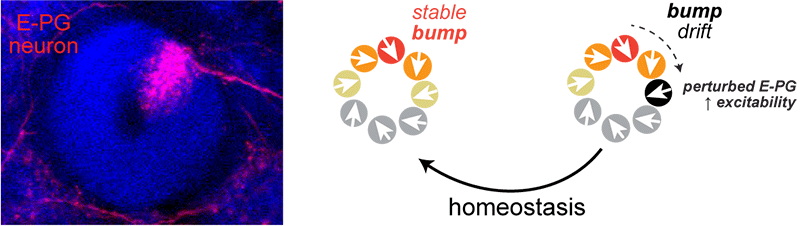
How are the population-level properties of neural networks maintained? The leading hypothesis for how information is held “in mind” during short-term memory is stably maintained neural activity. Within the ring-shaped compass network, the location of a “bump” of persistent neural activity stores a representation of the fly’s current heading direction. This means that in order to properly store information about which direction the fly is facing, the network must be balanced such that each location around the ring is equally favorable–this balance would be disrupted if the properties of individual neuronal elements ever changed. We are exploring how homeostatic regulation of single-cell synaptic and intrinsic properties shapes the compass network over the fly’s lifetime.
Selected Publications
Fisher, Y. E., Lu, J. D'Alessandro, I. Wilson, R. I. (2019) Sensorimotor experience remaps visual input to a heading-direction network. Nature 576, 121-125.
Isaacman-Beck, J. Paik, K. C., Wienecke, C. F. R., Yang, H. H., Fisher, Y. E., Wang, I. E., Ishida, I. G. Maimon, G. Wilson, R. I. Clandinin, T. R. (2020) SPARC enables genetic manipulation of precise proportions of cells. Nature Neuroscience. doi:10.1038/s41593-020-0668-9s
Fisher, Y. E.*, Yang, H. H.*, Isaacman-Beck, J., Xie, M., Gohl, D. M., Clandinin, T. R. (2017) FlpStop, a tool for conditional gene control in Drosophila. ELife 6: e22279 * equal contributions
Fisher, Y. E.*, Leong, J. C. S.*, Sporar, K., Ketkar, M. D., Gohl, D. M., Clandinin, T. R., Silies, M. (2015) A Class of Visual Neurons with Wide-Field Properties Is Required for Local Motion Detection. Current Biology, 25(3178-3189) * equal contributions
Fisher, Y. E.*, Silies, M.*, Clandinin, T. R. (2015) Orientation Selectivity Sharpens Motion Detection in Drosophila. Neuron 88 (390-402) * equal contributions
Silies, M. S.* Gohl, D.*, Fisher, Y. E., Freifeld, L., Clark, D., Clandinin, T. (2013) Modular Use of Peripheral Input Channels Tunes Motion-Detecting Circuitry. Neuron, 79(1), 111–12 * equal contributions
Last Updated 2021-05-07