Alanna Schepartz
CZ and Irmgard Distinguished Chair of Chemistry, Professor of Molecular Therapeutics
Lab Homepage: https://schepartzlab.com/Research Interests
Professor Schepartz’s research group is interested in questions that span the chemistry-biology continuum. We seek to establish new knowledge about the chemistry of complex cellular processes and apply this knowledge to design or discover molecules–both small and large–with unique or useful properties. We apply the tools of organic synthesis, biochemistry, biophysics, and structural, molecular, and synthetic biology in our work. Current projects focus on (1) repurposing the ribosome to biosynthesize sequence-defined chemical polymers and polyketides; (2) exploring and improving novel tools for trafficking proteins to the cytosol and nucleus for therapeutic applications; (3) understanding the mechanism by which chemical information is transported through cellular membranes; and (4) developing new probes and fluorophores to image organelle dynamics at super-resolution for highly extended times and in multiple colors.
Current Projects
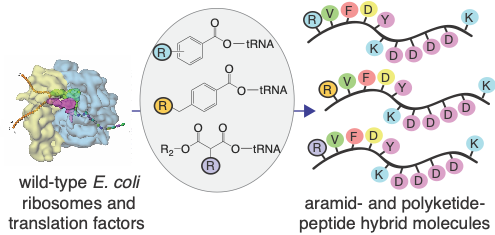 Repurposing the ribosome to synthesize sequence-defined chemical polymers. Polymers of one form or another power all living organisms and many technologies that advance science and technology. But today, our ability to rationally design and evolve new polymers is limited to the few natural macromolecules for which Nature has provided a genetic code. This limitation precludes the rapid development of next-generation materials, therapeutics, catalysts, fuels, and chemicals. Our group is part of a large and highly collaborative effort to expand the genetic code in an unprecedented way, by repurposing the bacterial translation apparatus to generate new, non-natural classes of sequence-defined, chemical polymers that bear only a glancing resemblance to natural macromolecules. Current work is focused on developing state-of-the-art chemical, synthetic, structural, and computational biology tools to enable, for the first time, the sequence-templated biosynthesis of aramids, polyolefins, polyurethanes, even polyketide precursors.
Repurposing the ribosome to synthesize sequence-defined chemical polymers. Polymers of one form or another power all living organisms and many technologies that advance science and technology. But today, our ability to rationally design and evolve new polymers is limited to the few natural macromolecules for which Nature has provided a genetic code. This limitation precludes the rapid development of next-generation materials, therapeutics, catalysts, fuels, and chemicals. Our group is part of a large and highly collaborative effort to expand the genetic code in an unprecedented way, by repurposing the bacterial translation apparatus to generate new, non-natural classes of sequence-defined, chemical polymers that bear only a glancing resemblance to natural macromolecules. Current work is focused on developing state-of-the-art chemical, synthetic, structural, and computational biology tools to enable, for the first time, the sequence-templated biosynthesis of aramids, polyolefins, polyurethanes, even polyketide precursors.
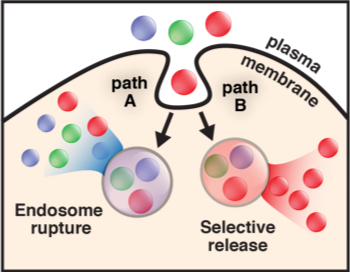 Improving the intracellular flow of therapeutic proteins. The inefficient delivery of proteins, peptides, and their mimetics into the mammalian cell cytosol limits their potential as therapeutics and research tools. Our laboratory discovered a family of cell-permeant miniature proteins (CPMPs) that are (1) non-toxic and (2) exploit a novel mechanism to traffic with unprecedented efficiency–with their cargo–into the cell cytosol and nucleus. Importantly, we have shown that the efficiency of transport into the cytosol and nucleus can be determined with accuracy and precision using fluorescence correlation spectroscopy (FCS). These studies have revealed that CPMPs are superior to every known cell-penetrating peptide examined, in large part because they more effectively access a previously unknown pathway for endosomal release that involves the HOPS complex, a natural endosome remodeling machine. Current work is focused on applying CPMPs to deliver therapeutic enzymes to offset difficult-to-treat inborn errors of metabolism, edit genes, or deliver antibody surrogates as protein-protein interaction inhibitors, as well as high-resolution structural and mechanistic studies to gain a deeper understanding of how CPMPs hijack the HOPS complex.
Improving the intracellular flow of therapeutic proteins. The inefficient delivery of proteins, peptides, and their mimetics into the mammalian cell cytosol limits their potential as therapeutics and research tools. Our laboratory discovered a family of cell-permeant miniature proteins (CPMPs) that are (1) non-toxic and (2) exploit a novel mechanism to traffic with unprecedented efficiency–with their cargo–into the cell cytosol and nucleus. Importantly, we have shown that the efficiency of transport into the cytosol and nucleus can be determined with accuracy and precision using fluorescence correlation spectroscopy (FCS). These studies have revealed that CPMPs are superior to every known cell-penetrating peptide examined, in large part because they more effectively access a previously unknown pathway for endosomal release that involves the HOPS complex, a natural endosome remodeling machine. Current work is focused on applying CPMPs to deliver therapeutic enzymes to offset difficult-to-treat inborn errors of metabolism, edit genes, or deliver antibody surrogates as protein-protein interaction inhibitors, as well as high-resolution structural and mechanistic studies to gain a deeper understanding of how CPMPs hijack the HOPS complex.
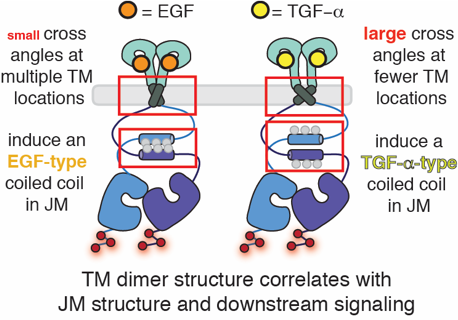 How do proteins encode and decode chemical information? We are very interested in the molecular processes by which proteins control the flow of chemical information through cellular membranes. In recent years we have exploited the virtues of the fluorogenic dye ReAsH to provide new insights into how EGFR communicates information across the plasma membrane; these insights have identified a potential therapeutic strategy to inhibit drug-resistant forms of EGFR. We discovered that the binding of most EGFR-specific growth factors to the receptor extracellular domain (ECD) induce the formation of one of two antiparallel coiled coils in the cytoplasmic juxtamembrane segment (JM) located hundreds of amino acids away. We subsequently discovered that JM coiled coil identity tracks with growth-factor dependent signaling, is influenced by activating mutations in the kinase domain and their inhibition status, and can be targeted by novel, allosteric, cell-permeable EGFR inhibitors. Most recently we discovered that the two JM coiled coils contain all the information necessary to direct EGFR into degradative or recycling endosomes: the JM functions as a traffic cop. Current work is focused on precisely how JM coiled coil structure is decoded to alter trafficking and downstream signaling, how this property can be exploited to deliver drug-resistant EGFR into lysosomes for degradation, and how analogous structures might be utilized for biased signaling by other kinases and GPCRs.
How do proteins encode and decode chemical information? We are very interested in the molecular processes by which proteins control the flow of chemical information through cellular membranes. In recent years we have exploited the virtues of the fluorogenic dye ReAsH to provide new insights into how EGFR communicates information across the plasma membrane; these insights have identified a potential therapeutic strategy to inhibit drug-resistant forms of EGFR. We discovered that the binding of most EGFR-specific growth factors to the receptor extracellular domain (ECD) induce the formation of one of two antiparallel coiled coils in the cytoplasmic juxtamembrane segment (JM) located hundreds of amino acids away. We subsequently discovered that JM coiled coil identity tracks with growth-factor dependent signaling, is influenced by activating mutations in the kinase domain and their inhibition status, and can be targeted by novel, allosteric, cell-permeable EGFR inhibitors. Most recently we discovered that the two JM coiled coils contain all the information necessary to direct EGFR into degradative or recycling endosomes: the JM functions as a traffic cop. Current work is focused on precisely how JM coiled coil structure is decoded to alter trafficking and downstream signaling, how this property can be exploited to deliver drug-resistant EGFR into lysosomes for degradation, and how analogous structures might be utilized for biased signaling by other kinases and GPCRs.
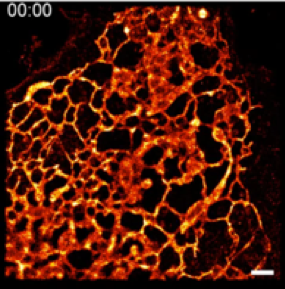 HIDE probes for long time-lapse imaging of organelle dynamics at super-resolution. Living cells are complex and dynamic assemblies that sequester and orchestrate processes that enable growth, division, regulation, movement, and communication. Membrane-bound organelles such as the endoplasmic reticulum, mitochondria, plasma membrane, and others are integral to these processes, and their functions demand dynamic reorganization in both space and time. Visualizing organelle dynamics in live cells over long time periods, especially at the nanoscale, has been almost impossible. In collaboration with a trio of cell biologists at Yale, our group developed a toolkit of two-component chemical tools called HIDE probes that allow scientists to visualize multiple organelles in live cells at super-resolution for unprecedented times. HIDE probes consist of two parts: an organelle-specific lipid or small molecule fused to a reactive functional group such as trans-cyclooctene (TCO) and a silicon-rhodamine dye (SiR or HMSiR) fused to the appropriate reaction partner. These parts, when added sequentially to cultured or primary cells, undergo an in situ click reaction that localizes the SiR dye within the organelle membrane. Here the photostability of the SiR chromophore is dramatically enhanced, generating images that last up to 50 times longer than those obtained with SiR-protein fusions. The first HIDE probes facilitate the visualization of the ER, mitochondria, and plasma membrane; more recent probes visualize late endosomes. Most recently, we developed strategies that support two-color HIDE imaging using both STED and SMS. Importantly, as HIDE probes do not require transfection, they can be applied readily by non-experts to image both primary and cultured cells. Current work is focused on expanding the HIDE toolbox to include additional colors and organelles, applying the tools that exist now to study organelle dynamics and interactions in disease, and exploiting what we have learned to develop HIDE probes for proteins.
HIDE probes for long time-lapse imaging of organelle dynamics at super-resolution. Living cells are complex and dynamic assemblies that sequester and orchestrate processes that enable growth, division, regulation, movement, and communication. Membrane-bound organelles such as the endoplasmic reticulum, mitochondria, plasma membrane, and others are integral to these processes, and their functions demand dynamic reorganization in both space and time. Visualizing organelle dynamics in live cells over long time periods, especially at the nanoscale, has been almost impossible. In collaboration with a trio of cell biologists at Yale, our group developed a toolkit of two-component chemical tools called HIDE probes that allow scientists to visualize multiple organelles in live cells at super-resolution for unprecedented times. HIDE probes consist of two parts: an organelle-specific lipid or small molecule fused to a reactive functional group such as trans-cyclooctene (TCO) and a silicon-rhodamine dye (SiR or HMSiR) fused to the appropriate reaction partner. These parts, when added sequentially to cultured or primary cells, undergo an in situ click reaction that localizes the SiR dye within the organelle membrane. Here the photostability of the SiR chromophore is dramatically enhanced, generating images that last up to 50 times longer than those obtained with SiR-protein fusions. The first HIDE probes facilitate the visualization of the ER, mitochondria, and plasma membrane; more recent probes visualize late endosomes. Most recently, we developed strategies that support two-color HIDE imaging using both STED and SMS. Importantly, as HIDE probes do not require transfection, they can be applied readily by non-experts to image both primary and cultured cells. Current work is focused on expanding the HIDE toolbox to include additional colors and organelles, applying the tools that exist now to study organelle dynamics and interactions in disease, and exploiting what we have learned to develop HIDE probes for proteins.
Selected Publications
Translation of diverse aramid- and 1,3-dicarbonyl-peptides by wild type ribosomes in vitro. O. Ad, K. Hoffmann, A. Cairns, A. Featherston, S.J. Miller, D. Söll, & A. Schepartz, ACS Cent. Sci. 2019, 5, 1289-1294.
HOPS-dependent endosomal fusion required for efficient cytosolic delivery of therapeutic peptides and small proteins. A. Steinauer, J.R. LaRochelle, R.F. Wissner, S. Berry, & A. Schepartz, Proc Natl Acad Sci USA 2019, 116, 512.
Mechanism of allosteric coupling into and through the plasma membrane by EGFR. J.K.L. Sinclair, A.S. Walker, A.E. Doerner, & A. Schepartz, Cell Chem. Biol. 2018, 857.
Long time-lapse nanocopy with spontaneously blinking membrane probes, Nature Biotechnology. H. Takakura, Y. Zhang, R.S. Erdmann, A.D. Thompson, Y. Lin, B. McNellis, F. Rivera-Molina, S.N. Uno, M. Kamiya, Y. Urano, J.E. Rothman, J. Bewersdorf, A. Schepartz, & D. Toomre, Nat. Biotechnol. 2017, 35, 773-780.
Last Updated 2019-09-12