New Faculty Profile | Molly Ohainle
The Secret Lives of Viruses
By Kirsten Mickelwait

Four decades after its initial discovery, the HIV virus is still an enormously important global human pathogen — there are many millions of people still being infected, with no vaccine or real cure in sight.
That’s one of the reasons Molly Ohainle studies HIV. The other is that it provides a perfect example of how a virus infects cells, especially one that has successfully jumped a species barrier. “We think that SARS-CoV-2 (COVID) has an animal origin,” she says. “And we know that HIV evolved from a related virus in primates, so it’s a virus that has evolved in a primate host and has only recently become successful at infecting human cells.”
When Ohainle joins MCB in July as assistant professor, she will continue her research in virology. Her lab will use high-throughput functional genomics approaches and molecular virology to study how cells are protected from viral infection. Within our cells, she explains, we express proteins that can block viral infection. But viruses are innovative and can devise ways to evade and work around these barriers, which makes the virus successful at infecting a host cell. Her lab will find and study how these processes work: both the cell’s defenses and the viruses’ ability to evade defenses.
“Say we have a few different defenses that our cells use to block virus infection, but viruses may have come up with 100 different ways of picking those locks,” Ohainle says. “Those are what I think are most exciting to study — all the ways that biology has devised to inhibit viral infection and how viruses cross these barriers to infection. I'm always amazed by how varied the strategies can be.”
Before coming to Cal, Ohainle spent seven years as a senior staff scientist at the Fred Hutchinson Cancer Research Center. There she had a breakthrough: developing an HIV-specific method of using CRISPR knockout technology to simultaneously look at many genes that are important for infection. “Now, in a single experiment and one flask of cells, we can examine thousands of genes and what effect all of those genes might have on virus replication,” she says.
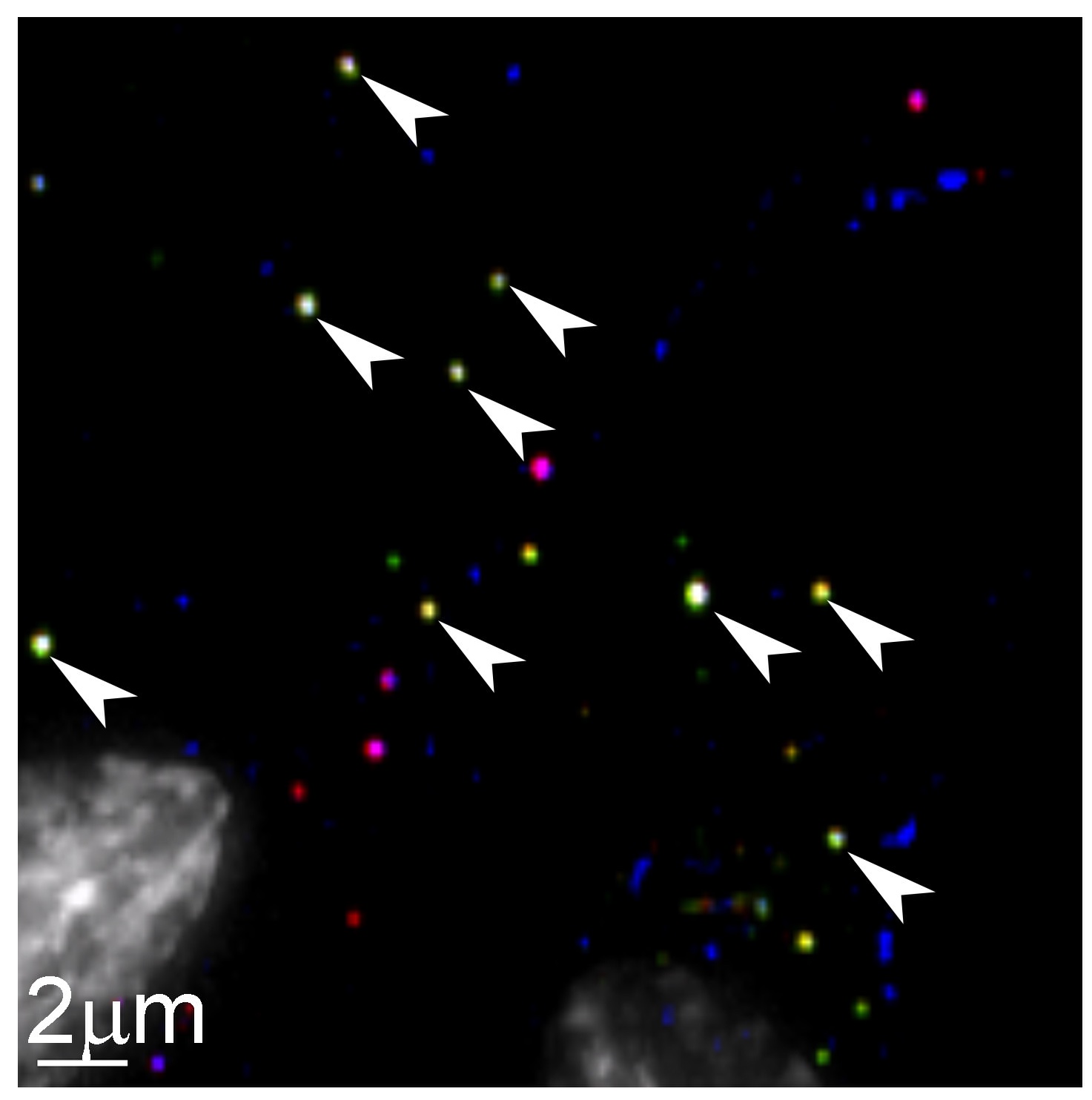
Currently, there are several potential applications for such technology. First is the possibility that human cellular responses could be altered such that a virus could no longer reactivate. A second would be drug discovery, targeting viral pathways to suppress the virus’s particular characteristics. And a third might be a clearer understanding of species barriers. “Because we've been infected by viruses throughout our evolution, they're one of the strongest selective pressures on our genome,” Ohainle says. “So there's this huge amount of diversity in sequence in antiviral genes among us and related animals.”
Ohainle’s earlier work explored how viral genetics and population immunity determine disease outcomes in dengue virus infection. When, during her postdoc, she gave birth to twins, with another young child already at home, she decided to put her academic research career on pause for a few years while she had three children under the age of three. Finding her own path back into research shapes Ohainle’s view on the importance of more inclusive policies and mentorship in science.
Now she looks forward to the possibility of collaboration with faculty across campus, who are exploring questions of how biology works at the cellular level. And she’s excited about bringing her high-throughput, deep-sequencing-based approaches back to a seminal location in the discovery of CRISPR technology.
More than anything, though, Ohainle looks forward to mentoring. “My best days are spent talking about data with my trainees—troubleshooting experiments, talking over new ideas,” she says. And she’s committed to making science more inclusive. “I know that Berkeley is trying hard to move in that direction, so I can't wait to join in that effort and push the institution to do better.”
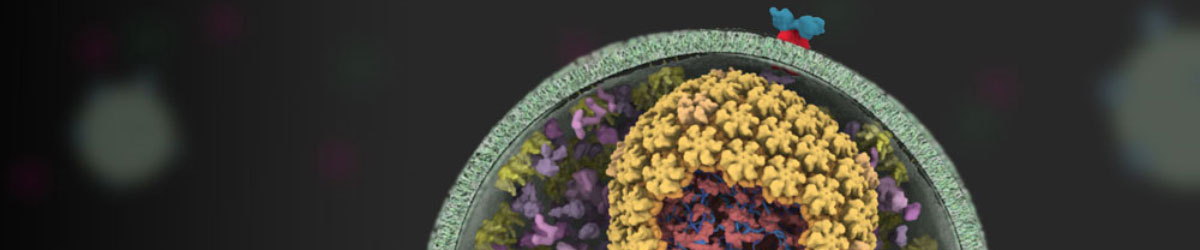
Banner image: Animation of the HIV life cycle. Image credit Janet Iwasa (Univ. of Utah) and scienceofhiv.org
Learn more about research in the Ohainle Lab at: https://theohainlelab.com/ or follow Molly Ohainle on Twitter @MollyOhainle.
Back to Main Spring 2022 Newsletter Page
| Connect With Us! | ||||
MCB Twitter |
 LinkedIn Postdocs, PhDs, or Undergrads |
 Cal Alumni Network |
 Give to MCB |
|
