Ellen Robey

IMM Division Head and
Professor of Immunology and Molecular Medicine*
*And Affiliate, Division of Cell and Developmental Biology
Research Interests
Our lab is interested in how signaling pathways control cell fate decisions. By using T cell development and immune responses in the mouse as model systems, we can take advantage of the powerful genetic approaches available in the mouse, while learning about a mammalian immune system that is very close to our own. Our lab also makes extensive use of 2-photon imaging approaches to observe and analyze T cell behavior in real-time in situ.
Current Projects
The control of T cell fate in the thymus. During thymic development, T cell precursors migrate to the thymus where they proliferate, rearrange their antigen receptor genes, and eventually give rise to the mature T cell subsets. During this process, the thymocytes are subject to a selection process that results in the death of ~99% of the cells and that shapes the mature T cell repertoire. We are investigating the mechanism that lead to positive and negative selection of T cells in the thymus, as well as the signaling events that control to cell fate decisions, including the CD4 versus CD8 cell lineage choice.
Immune responses to parasitic infection. Our lab is investigating host-pathogen interactions using a mouse infection model of the intracellular protozoan parasite, Toxoplasma gondii. We have established mouse infection models that enable us to quantitiate immune responses to the parasites in vivo and to visualize immune responses in real-time. Our ongoing efforts in this area are focused on examining CD8 T cell responses during priming and effector phases of the immune response, and to examining immune protection during chronic infection. For additional info go to http://mcb.berkeley.edu/chpr/
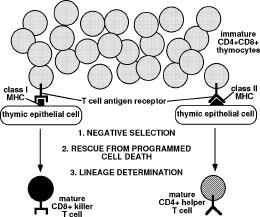
During T cell development in the thymus, thymocyte precursors that express both CD4 and CD8 undergo a process of cellular selection and lineage commitment. Potentially auto-reactive T cells whose antigen receptors can interact strongly with self are eliminated through negative selection. Thymocytes that lack antigen receptors, or whose antigen receptors cannot interact with MHC ligands are eliminated by programmed cell death. Thymocytes whose antigen receptors have weak reactivity to class I MHC are selected to develop into mature CD8+ T cells. Thymocytes whose antigen receptors have weak reactivity to class II MHC are selected to develop into mature CD4 lineage T cells.
Selected Publications
Lutes LK, Steier Z, McIntyre LL, Pandey S, Kaminski J, Hoover AR, Ariotti S, Streets A, Yosef N, Robey EA.Elife. (2021). T cell self-reactivity during thymic development dictates the timing of positive selection. Apr 22;10:e65435. doi: 10.7554/eLife.65435.
Tsitsiklis, A., Bangs, D. J., Lutes, L. K., Chan, S. W., Geiger, K. M., Modzelewski, A. J., , L. Labarta-Bajo, Y. Wang, E. I. Zuniga, S. Dai, and E. A. Robey. (2020). An Unusual MHC Molecule Generates Protective CD8+ T Cell Responses to Chronic Infection. Frontiers in Immunology, 11, 1464. http://doi.org/10.3389/fimmu.2020.01464.
Kurd, N. S., Hoover, A., Yoon, J., Weist, B. M., Lutes, L., Chan, S. W., & Robey, E. A. (2020). Factors that influence the thymic selection of CD8αα intraepithelial lymphocytes. Mucosal Immunology, 16(1), 355–12. http://doi.org/10.1038/s41385-020-0295-5.
Kurd, N. S., Lutes, L. K., Yoon, J., Chan, S. W., Dzhagalov, I. L., Hoover, A. R., & Robey, E. A. (2019). A role for phagocytosis in inducing cell death during thymocyte negative selection. eLife, 8, 1879. http://doi.org/10.7554/eLife.48097
Klein, L., Robey, E. A., & Hsieh, C.-S. (2018). Central CD4+ T cell tolerance: deletion versus regulatory T cell differentiation. Nature Reviews. Immunology, 19(1), 1–12. http://doi.org/10.1038/s41577-018-0083-6
Chu, H.H., Chan, S.W., Gosling, J.P., Blanchard, N., Tsitsiklis, A., Lythe, G., Shastri, N., Molina-París, C., and Robey, E.A. (2016). Continuous Effector CD8(+) T Cell Production in a Controlled Persistent Infection Is Sustained by a Proliferative Intermediate Population. Immunity 45, 159–171.
Weist, B.M., Kurd, N., Boussier, J., Chan, S.W., and Robey, E.A. (2015). Thymic regulatory T cell niche size is dictated by limiting IL-2 from antigen-bearing dendritic cells and feedback competition. Nat. Immunol. 16, 635–641.
Au-Yeung, B.B., Melichar, H.J., Ross, J.O., Cheng, D.A., Zikherman, J., Shokat, K.M., Robey, E.A., and Weiss, A. (2014). Quantitative and temporal requirements revealed for Zap70 catalytic activity during T cell development. Nat. Immunol. 15, 687–694.
Melichar, H.J., Ross, J.O., Herzmark, P., Hogquist, K.A., and Robey, E.A. (2013). Distinct temporal patterns of T cell receptor signaling during positive versus negative selection in situ. Science Signaling 6, ra92.
Germain, R.N., Robey, E.A., and Cahalan, M.D. (2012). A decade of imaging cellular motility and interaction dynamics in the immune system. Science
Photo Credit: Mark Hanson of Mark Joseph Studios
Last Updated 2021-08-05