Evoked and spontaneous transmission favored by distinct sets of synapses
Abstract
Spontaneous “miniature” transmitter release takes place at low rates at all synapses. Long thought of as an unavoidable leak, spontaneous release has recently been suggested to be mediated by distinct pre- and postsynaptic molecular machineries and to have a specialized role in setting up and adjusting neuronal circuits. It remains unclear how spontaneous and evoked transmission are related at individual synapses, how they are distributed spatially when an axon makes multiple contacts with a target, and whether they are commonly regulated.
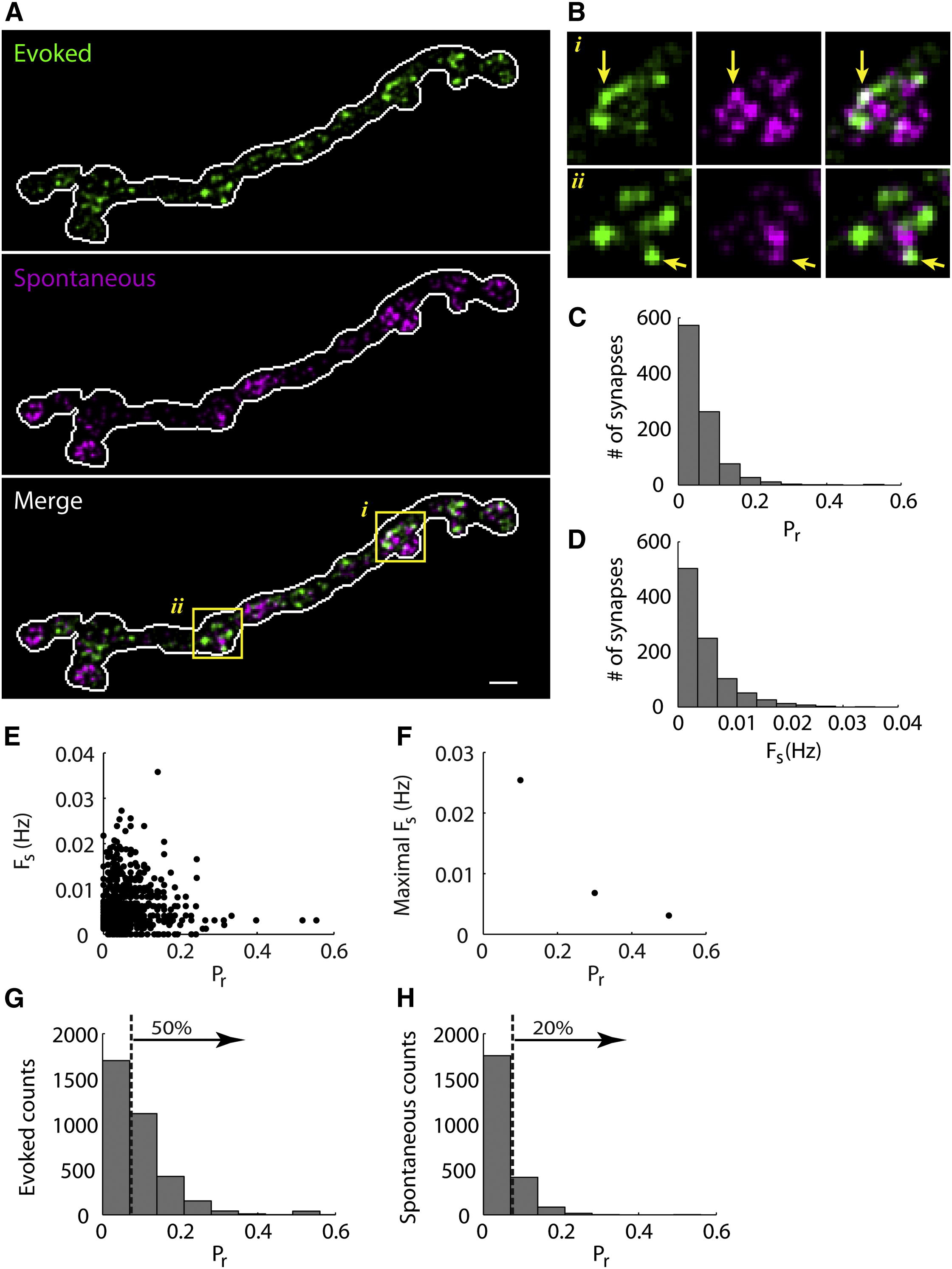
Electrophysiological recordings in the Drosophila larval neuromuscular junction, in the presence of the use-dependent glutamate receptor (GluR) blocker philanthotoxin, indicated that spontaneous and evoked transmission employ distinct sets of GluRs. In vivo imaging of transmission using synaptically targeted GCaMP3 to detect Ca2+ influx through the GluRs revealed little spatial overlap between synapses participating in spontaneous and evoked transmission. Spontaneous and evoked transmission were oppositely correlated with presynaptic levels of the protein Brp: synapses with high Brp favored evoked transmission, whereas synapses with low Brp were more active spontaneously. High-frequency stimulation did not increase the overlap between evoked and spontaneous transmission, and instead decreased the rate of spontaneous release from synapses that were highly active in evoked transmission.
Although individual synapses can participate in both evoked and spontaneous transmission, highly active synapses show a preference for one mode of transmission. The presynaptic protein Brp promotes evoked transmission and suppresses spontaneous release. These findings suggest the existence of presynaptic mechanisms that promote synaptic specialization to either evoked or spontaneous transmission.